400042 重庆,第三军医大学大坪医院野战外科研究所一室,创伤、烧伤与复合伤国家重点实验室2;
400010 重庆,重庆医科大学附属第二医院肝胆外科3
2State Key Laboratory of Trauma, Burns and Combined Injury, Department 1, Institute of Surgery Research, Daping Hospital, Third Military Medical University, Chongqing, 400042;
3Department of Hepatobiliary Surgery, the Second Affiliated Hospital of Chongqing Medical University, Chongqing, 400010, China
感染、创伤、烧伤、缺血再灌注等均可引起全身性炎症反应综合征(systemic inflammatory response syndrome,SIRS)。炎症反应表现为炎症因子增加、细胞死亡、组织损伤等,若患者未得到有效治疗,最后发展为多器官功能障碍综合征,导致其死亡[1, 2]。内毒素脂多糖是革兰阴性(G-)菌细胞壁的主要成分,是导致SIRS的强烈致炎物质。由LPS诱导的炎症因子过度释放,最终导致严重脓毒症和败血症休克等临床症状[3, 4, 5]。胰岛素样生长因子-1受体(insulin-like growth factor 1 receptor,IGF-1R)在炎症的发生、发展过程中发挥重要作用。IGF-1R磷酸化诱导下游PI3K/Akt和NF-κB信号通路活化,导致促炎因子过度释放和组织损伤等炎症反应[6]。PQ401是IGF-1R的选择性抑制剂,通过抑制IGF-1R自身磷酸化,发挥诸多生物学效应[7, 8]。本研究团队在研究吴茱萸碱调节炎性巨噬细胞IGF-1受体磷酸化的同时,发现PQ401具有潜在的抗炎作用。本研究探讨PQ401的体内外抗炎效应,以证实PQ401对脓毒症具有治疗作用。
1 材料与方法 1.1 实验动物与试剂7周龄清洁级雄性C57BL/6小鼠50只,体质量19~22 g,购自第三军医大学大坪医院实验动物中心,动物合格证号:SCXX(军)2012-008。PQ401购自美国Selleck公司(PQ401溶于DMSO),LPS购自美国Sigma公司,小鼠IL-1β、IL-6、TNF-α酶联免疫吸附实验(ELISA)试剂盒及细胞质、细胞核提取试剂盒均购自武汉博士德生物工程有限公司,核转录因子-κB(NF-κB)p65抗体购自美国Santa Cruz公司,核内参TATA结合蛋白(TBP)购自美国Abcam公司,RPMI1640培养基购自美国Invitrogen公司,胎牛血清购自美国Gibco公司,RAW264.7细胞株为本实验室保存。
1.2 动物实验将50只小鼠按随机数字表法分为对照组、模型组和不同剂量PQ401组,每组10只。腹腔注射LPS 10 mg/kg 制备急性炎症反应小鼠模型;对照组腹腔注射等量二甲基亚砜(DMSO);PQ401组分别腹腔注射LPS 10 mg/kg和PQ401 25、50、100 mg/kg。于建模12 h 后处死小鼠,取血和肝脏组织备检。本实验动物处置方法符合动物伦理学标准。
1.3 ELISA检测细胞上清TNF-α、IL-1β、IL-6水平将RAW264.7细胞铺于24孔板中,预先用0.1、1、10 μmol/L PQ401作用1 h,然后加入10 μg/mL LPS作用12 h,收取细胞上清备检。另将RAW264.7细胞 预先用10 μmol/L PQ401作用1 h,然后加入10 μg/mL LPS作用6、12、24 h,收取细胞上清备检。LPS对照组在作用6 h后收取细胞上清。严格按照ELISA试剂盒说明书进行细胞因子测定。
1.4 ELISA检测血清、肝组织匀浆TNF-α、IL-1β、IL-6水平腹腔注射12 h后摘除小鼠眼球放血,静置1 h后离心取血清备检。同时取肝脏组织,加入蛋白裂解液,离心取上清备检。严格按照ELISA试剂盒说明书进行细胞因子测定。
1.5 Western blot检测肝细胞核内NF-κB p65蛋白表达建模后12 h取小鼠肝脏组织,冰上研磨剪碎,提取胞核蛋白,测定蛋白浓度,采用Western blot检测NF-κB p65蛋白表达。以目的蛋白与核内参TBP的灰度值比值表示蛋白表达量。
1.6 肝功能指标检测建模后12 h摘除小鼠眼球取血,静置1 h后离心取血清,使用全自动生化分析仪检测丙氨酸转氨酶(ALT)、天冬氨酸转氨酶(AST)、总胆红素(TBil)含量。
1.7 统计学分析采用SPSS 18.0统计软件进行单因素方差分析和LSD-t检验,结果以x±s表示。
2 结果 2.1 PQ401对LPS刺激RAW264.7细胞上清TNF-α、IL-1β、IL-6水平的影响在LPS刺激RAW264.7细胞后,LPS组细胞上清TNF-α、IL-1β、IL-6水平均明显高于正常对照组(P<0.05);经不同浓度和不同时间PQ401作用后,上清中的TNF-α、IL-1β、IL-6水平明显低于LPS组(P<0.05),呈浓度和时间依赖性降低(表 1、2)。
| (μg/L,n=8,x±s) | |||
| 组别 | TNF-α | IL-1β | IL-6 |
| 对照组 | 2.6±0.3 | 3.9±0.2 | 1.2±1.1 |
| LPS组 | 43.7±3.1a | 57.7±2.1a | 13.2±1.2a |
| 0.1 μmol/L PQ401组 | 33.2±4.6ab | 39.6±5.2ab | 7.4±0.7ab |
| 1 μmol/L PQ401组 | 29.5±3.2ab | 34.7±6.2ab | 6.6±0.7ab |
| 10 μmol/L PQ401组 | 22.7±2.7ab | 29.3±5.3ab | 5.6±0.9ab |
| a:P<0.05,与对照组比较;b:P<0.05,与LPS组比较 | |||
| (μg/L,n=8,x±s) | |||
| 组别 | TNF-α | IL-1β | IL-6 |
| 对照组 | 2.1±0.4 | 2.4±0.5 | 1.5±1.4 |
| LPS组 6 h | 27.6±1.7a | 38.0±2.8a | 8.6±0.7a |
| 10 μmol/L PQ401组6 h | 13.6±2.1ab | 15.7±4.9ab | 3.7±0.6ab |
| LPS组 12 h | 54.2±2.5a | 60.5±3.2a | 15.2±0.9a |
| 10 μmol/L PQ401组 12 h | 31.6±3.2ab | 40.8±3.4ab | 6.2±0.4ab |
| LPS组 24 h | 110.3±4.8a | 125.5±4.8a | 35.4±0.7a |
| 10 μmol/L PQ401组 24 h | 58.7±3.7ab | 67.1±5.2ab | 19.6±0.6ab |
| a:P<0.05,与对照组比较;b:P<0.05,与对应时相点LPS组比较 | |||
对照组小鼠精神状态良好,活跃,正常进食、饮水。模型组小鼠在建模1 h后出现寒颤、体温降低、聚集成群、腹泻、进食和饮水减少等表现。PQ401组小鼠表现明显好于模型组,比对照组小鼠状态差。
2.3 各组小鼠血清TNF-α、IL-1β、IL-6水平模型组血清TNF-α、IL-1β、IL-6水平均明显高于对照组(P<0.05);经PQ401治疗后血清TNF-α、IL-1β、IL-6水平明显低于模型组(P<0.05,表 3)。
| (n=10,x±s) | |||
| 组别 | TNF-α(μg/L) | IL-1β(μg/L) | IL-6(mg/L) |
| 对照组 | 56.3±2.4 | 0.2±0.1 | 0.1±0.1 |
| 模型组 | 526.2±36.7a | 205.4±12.2a | 21.7±2.0a |
| 25 mg/kg PQ401组 | 394.5±51.4ab | 154.5±14.5ab | 16.5±2.5ab |
| 50 mg/kg PQ401组 | 314.5±67.5ab | 106.5±24.8ab | 13.0±1.3ab |
| 100 mg/kg PQ401组 | 234.5±67.5ab | 84.5±67.5ab | 10.5±1.1ab |
| a:P<0.05,与对照组比较;b:P<0.05,与模型组比较 | |||
模型组肝组织TNF-α、IL-6水平及肝细胞核内NF-κB p65表达均显著高于对照组(P<0.01);但50 mg/kg PQ401组肝组织TNF-α、IL-6水平均显著低于模型组(P<0.05),PQ401组肝细胞核内NF-κB p65表达与模型组无显著区别(P>0.05,图 1、表 4)。
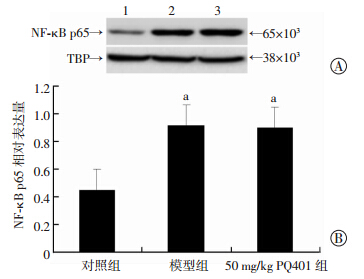 |
| A: Western blot检测 1:对照组;2: 模型组;3:50 mg/kg PQ401组; B:半定量分析 a: P<0.05,与对照组比较 图 1 Western blot检测各组小鼠肝细胞核内NF-κB的表达 |
| (n=10,x±s) | ||
| 组别 | TNF-α (ng/g) | IL-6 (pg/g) |
| 对照组 | 57.1±3.8 | 21.6±2.4 |
| 模型组 | 383.3±68.6a | 320.7±29.2a |
| 50 mg/kg PQ401组 | 232.2±31.1ab | 214.4±37.1ab |
| a:P<0.05,与对照组比较;b:P<0.05,与模型组比较 | ||
模型组血清ALT、AST、TBil水平均较对照组显著增加(P<0.05);50 mg/kg PQ401组血清ALT、AST、TBil水平与模型组比较差异无统计学意义(P>0.05,表 5)。
| (n=10,x±s) | |||
| 组别 | ALT(U/L) | AST(U/L) | TBil(μmol/L) |
| 对照组 | 23.7±1.9 | 103.6±5.6 | 3.8±0.2 |
| 模型组 | 165.1±18.6 a | 803.7±24.9a | 11.8±1.4a |
| PQ401组 | 154.9±6.9 a | 789.9±13.9a | 11.6±0.9 a |
| a:P<0.05,与对照组比较 | |||
PQ401是近年发现的选择性IGF-1R抑制剂,其本质为二芳基脲化合物[7]。IGF-1R抑制剂在体内发挥众多生物学效应,如对于非小细胞肺癌、卵巢癌、鳞状细胞癌、尤文氏肉瘤等肿瘤疾病有显著治疗作用[9, 10, 11, 12];IGF-1R抑制剂能缓解中枢体温调节紊乱引发的高热[13],但是PQ401的抗炎作用,至今尚不清楚。
在众多细胞实验中,小鼠单核/巨噬细胞株RAW264.7是建立体外炎症模型的重要工具[14, 15, 16, 17, 18]。由LPS刺激RAW264.7细胞后,脱氢木香内酯、阿伐他丁、壳聚糖等抗炎药物能显著降低炎症因子IL-6、IL-1β、TNF-α的释放[15, 18, 19]。在本研究中发现PQ401有效降低LPS诱导RAW264.7细胞炎症因子释放,有潜在的抗炎效应。
由LPS建立的小鼠脓毒症模型具有稳定性高、重复性好的特点,因此,其广泛应用于各项动物实验中[3]。脓毒症是重症监护病房(ICU)患者死亡的主要原因,而脓毒症的特点为全身急性炎症反应综合征,表现为炎症介质IL-6、IL-1β、TNF-α过度释放,因此,炎症是机体抵抗脓毒症过程中重要机制[14]。Kim等[20]发现,给予爆发性肝衰竭小鼠羽扇醇治疗后,IL-6、TNF-α降低,急性炎症得到有效逆转;有研究发现胍丁胺对脂多糖或酵母多糖诱导的急性炎症小鼠模型都有明显的保护效应,能明显抑制小鼠血清和肝脏中炎症因子释放[21, 22, 23];因此,抑制血清炎症因子释放和脏器巨噬细胞活化是发挥抗炎作用的重要指标。PQ401能显著降低血清和肝组织中的炎症因子TNF-α、IL-1β、IL-6的释放,可见,PQ401对LPS诱导的小鼠炎症模型有保护效应。
核转录因子NF-κB在LPS诱导的促炎信号通路中发挥重要作用[24, 25, 26]。Lee等[27]报道,Rubus coreanus通过抑制NF-κB和MAPK活化从而有效缓解慢性炎症反应;Finkin-Groner等[28]用氨基甲酸酯治疗LPS诱导的小鼠肺损伤,发现其通过p38 MAPK、AP-1 和 NF-κB信号通路发挥抗炎作用,可见药物可通过多条抗炎通路发挥抗炎作用。本实验中,PQ401虽然抑制LPS所致小鼠炎症反应,明显降低血清和肝脏中炎症因子的释放,但是却并未影响NF-κB信号通路,这表明PQ401的抗炎作用是通过非NF-κB信号通路来实现的。
综上所述,PQ401在体外可降低LPS诱导的RAW264.7细胞的炎症因子TNF-α、IL-1β、IL-6的分泌,同时在体内对LPS所引发的血清和肝组织的炎症因子均具有抑制作用,但是其作用机制与NF-κBp65无关,可能是通过其他的信号通路来发挥抗炎效应,此问题有待进一步研究。
| [1] | Cauwels A,Rogge E,Vandendriessche B,et al.Extracellular ATP drives systemic inflammation,tissue damage and mortality[J].Cell Death Dis,2014,5:e1102. |
| [2] | Louis K,Netea M G,Carrer D P,et al.Bacterial translocation in an experimental model of multiple organ dysfunctions[J].J Surg Res,2013,183(2):686-694. |
| [3] | Thomas R C,Bath M F,Stover C M,et al.Exploring LPS-induced sepsis in rats and mice as a model to study potential protective effects of the nociceptin/orphanin FQ system[J].Peptides,2014,61:56-60. |
| [4] | Plociennikowska A,Hromada-Judycka A,Borzecka K,et al.Co-operation of TLR4 and raft proteins in LPS-induced pro-inflammatory signaling[J].Cell Mol Life Sci,2015,72(3):557-581. |
| [5] | Anderson S T,Commins S,Moynagh P N,et al.Lipopolysaccharide-induced sepsis induces long-lasting affective changes in the mouse[J].Brain Behav Immun,2015,43:98-109. |
| [6] | Zhao D,Bakirtzi K,Zhan Y,et al.Insulin-like growth factor-1 receptor transactivation modulates the inflammatory and prolif0erative responses of neurotensin in human colonic epithelial cells[J].J Biol Chem,2011,286(8):6092-6099. |
| [7] | Gable K L,Maddux B A,Penaranda C,et al.Diarylureas are small-molecule inhibitors of insulin-like growth factor I receptor signaling and breast cancer cell growth[J].Mol Cancer Ther,2006,5(4):1079-1086. |
| [8] | Troib A,Landau D,Youngren J F,et al.The effects of type 1 IGF receptor inhibition in a mouse model of diabetic kidney disease[J].Growth Horm IGF Res,2011,21(5):285-291. |
| [9] | Jameson M J,Beckler A D,Taniguchi L E,et al.Activation of the insulin-like growth factor-1 receptor induces resistance to epidermal growth factor receptor antagonism in head and neck squamous carcinoma cells[J].Mol Cancer Ther,2011,10(11):2124-2134. |
| [10] | Ji Q S,Mulvihill M J,Rosenfeld-Franklin M,et al.A novel,potent,and selective insulin-like growth factor-I receptor kinase inhibitor blocks insulin-like growth factor-I receptor signaling in vitro and inhibits insulin-like growth factor-I receptor dependent tumor growth in vivo[J].Mol Cancer Ther,2007,6(8):2158-2167. |
| [11] | Manara M C,Landuzzi L,Nanni P,et al.Preclinical in vivo study of new insulin-like growth factor-I receptor:specific inhibitor in Ewing's sarcoma[J].Clin Cancer Res,2007,13(4):1322-1330. |
| [12] | Dong Z,Yao M,Wang L,et al.Abnormal expression of insulin-like growth factor-I receptor in hepatoma tissue and its inhibition to promote apoptosis of tumor cells[J].Tumour Biol,2013,34(6):3397-3405. |
| [13] | Sanchez-Alavez M,Osborn O,Tabarean I V,et al.Insulin-like growth factor 1-mediated hyperthermia involves anterior hypothalamic insulin receptors[J].J Biol Chem,2011,286(17):14983-14990. |
| [14] | Nguyen P H,Zhao B T,Lee J H,et al.Isolation of benzoic and cinnamic acid derivatives from the grains of Sorghum bicolor and their inhibition of lipopolysaccharide-induced nitric oxide production in RAW 264.7 cells[J].Food Chem,2015,168:512-519. |
| [15] | Zhu J,Zhang Y,Wu G,et al.Inhibitory effects of oligochitosan on TNF-alpha,IL-1beta and nitric oxide production in lipopolysaccharide-induced RAW264.7 cells[J].Mol Med Rep,2015,11(1):729-733. |
| [16] | Yin X,Gong X,Jiang R,et al.Emodin ameliorated lipopolysaccharide-induced fulminant hepatic failure by blockade of TLR4/MD2 complex expression in D-galactosamine-sensitized mice[J].Int Immunopharmacol,2014,23(1):66-72. |
| [17] | Ye M,Wang Q,Zhang W,et al.Oroxylin A exerts anti-inflammatory activity on lipopolysaccharide-induced mouse macrophage via Nrf2/ARE activation[J].Biochem Cell Biol,2014,92(5):337-348. |
| [18] | Wang X Q,Luo N S,Salah Z Q,et al.Atorvastatin Attenuates TNF-alpha Production via Heme Oxygenase-1 Pathway in LPS-stimulated RAW264.7 Macrophages[J].Biomed Environ Sci,2014,27(10):786-793. |
| [19] | Park E J,Park S W,Kim H J,et al.Dehydrocostuslactone inhibits LPS-induced inflammation by p38MAPK-dependent induction of hemeoxygenase-1 in vitro and improves survival of mice in CLP-induced sepsis in vivo[J].Int Immunopharmacol,2014,22(2):332-340. |
| [20] | Kim S J,Cho H I,Kim S J,et al.Protective effects of lupeol against D-galactosamine and lipopolysaccharide-induced fulminant hepatic failure in mice[J].J Nat Prod,2014,77(11):2383-2388. |
| [21] | 顾颖,范霞,张醇,等.胍丁胺对酵母多糖诱导急性肺损伤的器官保护作用[J].中国危重病急救医学,2011,23(11):665-668. |
| [22] | 李炫飞,范霞,郑志华,等.胍丁胺对脂多糖诱导急性肝损伤的保护作用[J].中华危重病急救医学,2013,25(12):720-724. |
| [23] | Li X,Liu Z,Jin H,et al.Agmatine protects against zymosan-induced acute lung injury in mice by inhibiting NF-kappaB-mediated inflammatory response[J].Biomed Res Int,2014,2014:583736. |
| [24] | Verstrepen L,Carpentier I,Beyaert R.The biology of A20-binding inhibitors of NF-kappaB activation (ABINs)[J].Adv Exp Med Biol,2014,809:13-31. |
| [25] | Yang C,Yu L,Kong L,et al.Pyrroloquinoline quinone (PQQ) inhibits lipopolysaccharide induced inflammation in part via downregulated NF-kappaB and p38/JNK activation in microglial and attenuates microglia activation in lipopolysaccharide treatment mice[J].PLoS One,2014,9(10):e109502. |
| [26] | Zhang Y,Liu C,Dong B,et al.Anti-inflammatory activity and mechanism of surfactin in lipopolysaccharide-activated macrophages[J].Inflammation,2015,38(2):756-764. |
| [27] | Lee J E,Cho S M,Park E,et al.Anti-inflammatory effects of Rubus coreanus Miquel through inhibition of NF-kappaB and MAP Kinase[J].Nutr Res Pract,2014,8(5):501-508. |
| [28] | Finkin-Groner E,Moradov D,Shifrin H,et al.Indoline-3-propionate and 3-aminopropyl carbamates reduce lung injury and pro-inflammatory cytokines induced in mice by LPS[J].Br J Pharmacol,2015,172(4):1101-1113. |



